Life of an Air Flask
NOAA Research, the research and development arm of NOAA, conducts research to provide the nation with better weather forecasts, earlier warnings for natural disasters, and an overall greater understanding of our oceans, climate, and atmosphere. This research helps prepare the nation for the new challenges of tomorrow as society and natural surroundings continue to change.
At a NOAA laboratory in Boulder, Colorado, flask-filled shelves line the walls. The lab is home to thousands of glass cylinders that travel the world to sample Earth’s air. None of the flasks rest on these shelves for long before stowing away for journeys to some of the world’s most remote locations. They are vital workhorses used to monitor the small fraction of the atmosphere containing trace levels of gases and particles that can have profound effects on the health and condition of our planet. This first “person” travelogue of "flask 3331" discusses the air sampling that enables NOAA to explain global climate and air quality as well some of the mundane, but necessary, “shipping and handling” considerations for running a worldwide atmospheric monitoring program.

The “author” of this story with some of approximately 6,000 flasks in NOAA’s Earth System Research Laboratory in Boulder, Colorado. The flasks travel to far-flung corners of the planet to be filled with air samples for global atmosperhic monitoring.
I was born in Boulder, Colorado. Not many can say that, as Boulder is a place that attracts a lot of imports – like college kids who don’t leave after graduation, outdoorsy types, and international scientists. It’s hard to find a native in Boulder.
Don’t think that I’ve just been hanging around here, though. I’ve been all over the world–inside the Arctic Circle in Alaska, the Caribbean island of Barbados (which I liked so much I went twice), across the Pacific on a cargo ship, and to Ascension Island.
Anyhow, I’ve been around, as they say. In just the past two years, I've ventured to the Tae-Ahn Peninsula in South Korea and Constanta, Romania, on the Black Sea. Early in 2007, I shipped out to Argentina, at the very tippy-tip of South America, on Tierra del Fuego. In between trips, I always come back to Boulder for a month or so, where I get analyzed, evacuated, and sent out again.
Let me explain...
Far Away Places
They call me #3331, but you can call me a NOAA air-sampling flask. I am one of about 6,000 containers used to collect air samples all over the world. The Global Monitoring Division, part of NOAA’s Earth System Research Laboratory in Boulder, runs the air sampling network and sends us air flasks to 55 locations around the globe. Some of us even go down to the South Pole and “winter over” there, when the sun doesn’t come up for six months.
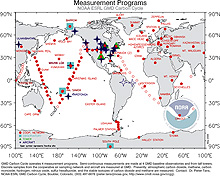
The 55 locations around the globe where volunteers take air samples every week and return them to NOAA in Boulder, Colorado, for analysis of trace gases. Click image for larger view.
Mostly it’s volunteers who carefully and consistently collect the air. Like the retired school teacher in the Gobi Desert (that’s in Mongolia). Or Oskar, the lighthouse keeper in Iceland. Or the first mate on a cargo ship to New Zealand. They all take two samples every week all over the world.
You’ll notice that we don’t go to New York City or Los Angeles, unless we’re just passing through. You won’t see us in Beijing or Bangkok or Buenos Aires. We travel to remote places, scientists are tracking the global mix of the atmosphere, not looking at local sources of pollution from major cities or industrial centers.
Measuring Trace Gases
Those of you who recall your science classes might remember that the composition of the air, generally speaking, is 21 percent oxygen and 78 percent nitrogen. The remaining one percent contains other gases in very small amounts. We’re helping to measure concentrations of these trace gases, some of which are greenhouse gases.
You’ve probably heard about greenhouse gases. They are gases like carbon dioxide, methane, and nitrous oxide. When they get into the atmosphere, they don’t allow as much heat, or infrared radiation, to escape from the Earth into space. So, these gases warm up the surface of the Earth. Some of this warming is needed (unless you would like an average temperature of zero degrees Fahrenheit!) But, too much greenhouse warming could tilt the global climate too far the other way, and that’s a possibility that concerns scientists.
Besides tracking trace gases like carbon dioxide and methane that cause warming of the planet, other gases are measured to follow the carbon cycle, to discover how carbon transfers among storage places in the oceans and in plants (which take in carbon dioxide as they grow) and gets into the atmosphere. Another gas, sulfur hexafluoride, is a greenhouse gas that is also measured so that scientists can trace fossil fuel emissions and track the movement of the atmosphere.
Sampling Around the Globe
So every week, in all 55 locations around the globe, someone collects two air samples at the same location on the same day of the week. Both samples are taken at the same time. If the concentrations of trace gases in one sample are very different from those in the other, scientists have to question if sampling was done correctly or ask what might have made two samples taken at the same place and time so different.
The people who take the samples use a case (about the size of a regular suitcase) with a battery-powered pump in it. They hook up two flasks and thrust a long pole with an intake hose a couple meters into the air so no one will taint the sample with carbon dioxide, the gas that humans exhale. When they turn on the pump, it flushes out the “synthetic air” that we flasks have been carrying, and fills us with about 2,500 cubic centimeters of air from the sampling location.
Traveling Back to Boulder

Most NOAA air sampling flasks travel humbly in cardboard boxes via parcel post. Click image for larger view.
Once they fill us with air, the samplers ship us back to Boulder. Did I tell you we are made of glass? It seems that no matter how well we’re packed, the shipping companies don’t always treat us as gently as they should. About two percent of us are damaged or broken every year. That can get expensive since we cost about $220 each! One time, an observant citizen visited the lab in Boulder with a few boxes he’d picked up on the road after they fell out of a truck. Due to international politics, some of us get shipped in a diplomatic pouch. But, mostly we just take our chances with something like parcel post.
Dr. Tom Conway, a research chemist at the lab who has been running this air sampling network for years, is always trying to figure out ways to better protect us. He came up with a new foam wrapping that stabilizes us in our boxes. And the newer flasks among us have a bright blue insulating material baked onto us when we first get to the lab. (The material on the older flasks is white.) Nevertheless, Tom jokes that in his lab, “ ‘PhD’ means packing, handling, and delivery.”
For reasons unknown, some of us never make it back to Boulder. Tom got an e-mail not long ago from a lieutenant in the U.S. Air Force. The airman wanted to know what he should do with several boxes that were sitting around Lajes Field, an airbase in the Azores. Tom had wondered why the air samples from that location weren’t coming in, but finally, several months’ worth of samples made their way back to the lab. Others never do.
Molly’s Lab

Kelly Sours fits air flasks on a carousel for automated analysis of the air sample. Click image for larger view.
Once back home, in Boulder, we go first to Molly’s lab. All of us know Molly Heller, because we pass through her lab both coming and going out of Boulder. She checks us in and sends us upstairs for our air to be analyzed. Up there, someone puts us on a carousel, makes sure the computer knows who we are and where we’ve been. Next, the automatic analysis begins. The scientists measure levels of carbon dioxide by using an instrument that analyzes the infrared absorption of the air inside us. (Remember, that’s how greenhouse gases are characterized—by their absorption of infrared radiation.)
Some of us flasks occasionally find our way to NOAA’s Mauna Loa Observatory, high on a mountain on the island of Hawaii. Here, we help to add to a long-term data set on carbon dioxide, a trace gas that makes up only 0.04 percent of the Earth’s atmosphere.
Molly then sends us over to a nearby laboratory at the University of Colorado that breaks down the samples even more to determine the isotopic ratios of the gases. That’s a fancy way of saying that atmospheric scientists can figure out whether the carbon dioxide came from sources on the land or sources in the sea. Knowing the source of carbon dioxide is important because it may one day help in managing the output of this gas.

Flasks being cleaned and prepared to be sent out again in the Boulder laboratory.
Back we go to Molly, who cleans us up to go out again. That’s why I am waiting for evacuation now. That is, we need to have the previous sample that’s been analyzed totally evacuated or cleaned out of us, and then we get tested for leaks or damage.
When I get back from my next trip, and my air sample is analyzed, Molly will examine and evacuate me, put me back in a box and ship me out again, with my mate, #3332. (We really do travel in pairs!) I won’t know where #3332 and I are going until the person at our destination opens the box. It might be one of the new sites we’re working on, like along the Gulf Coast of Mexico or on Taiwan. Anywhere is fine with me; I’m just happy to be part of NOAA’s careful, consistent, and continual worldwide air sampling network that shows scientists the composition of the atmosphere. Tracking trace gases in the atmosphere can help to explain warming of the climate, the movement of the atmosphere, and the source of greenhouse gases.
Contributed by Carol Knight, NOAA’s Office of Oceanic and Atmospheric Research
